Confocal, Super-Resolution & Live-Cell
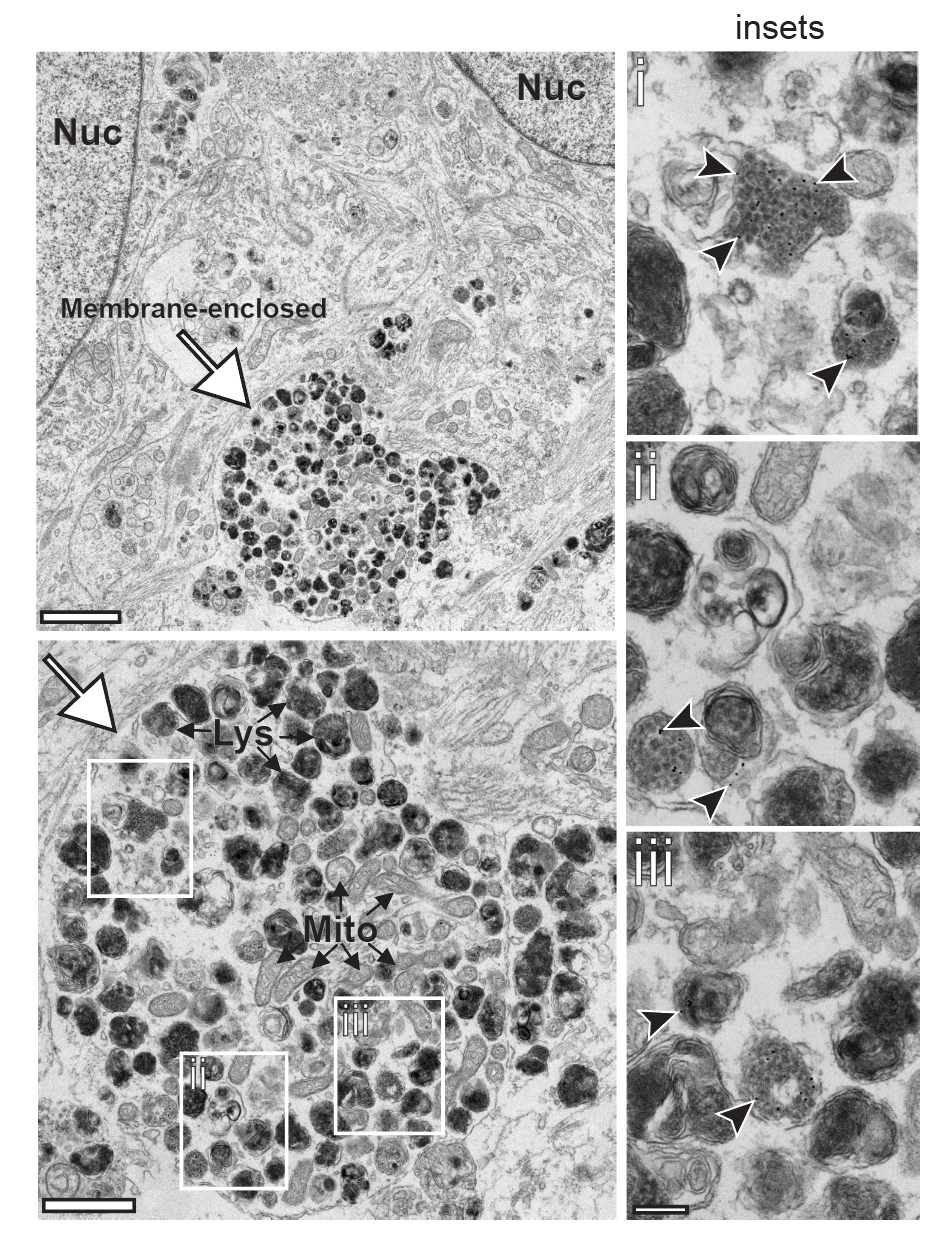
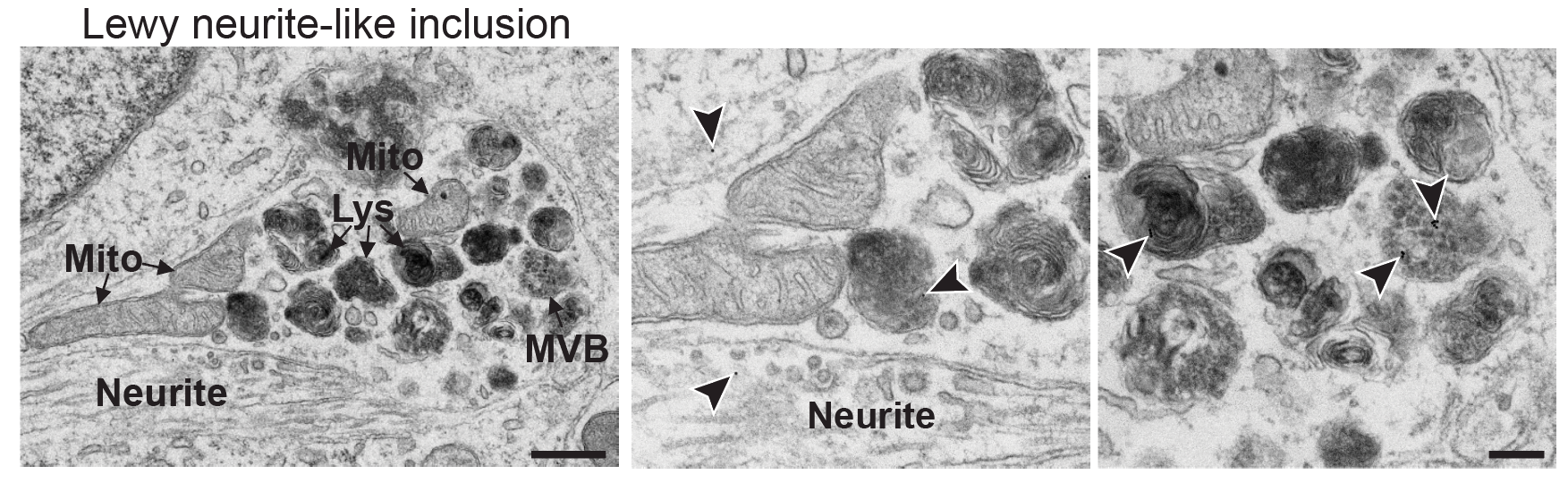
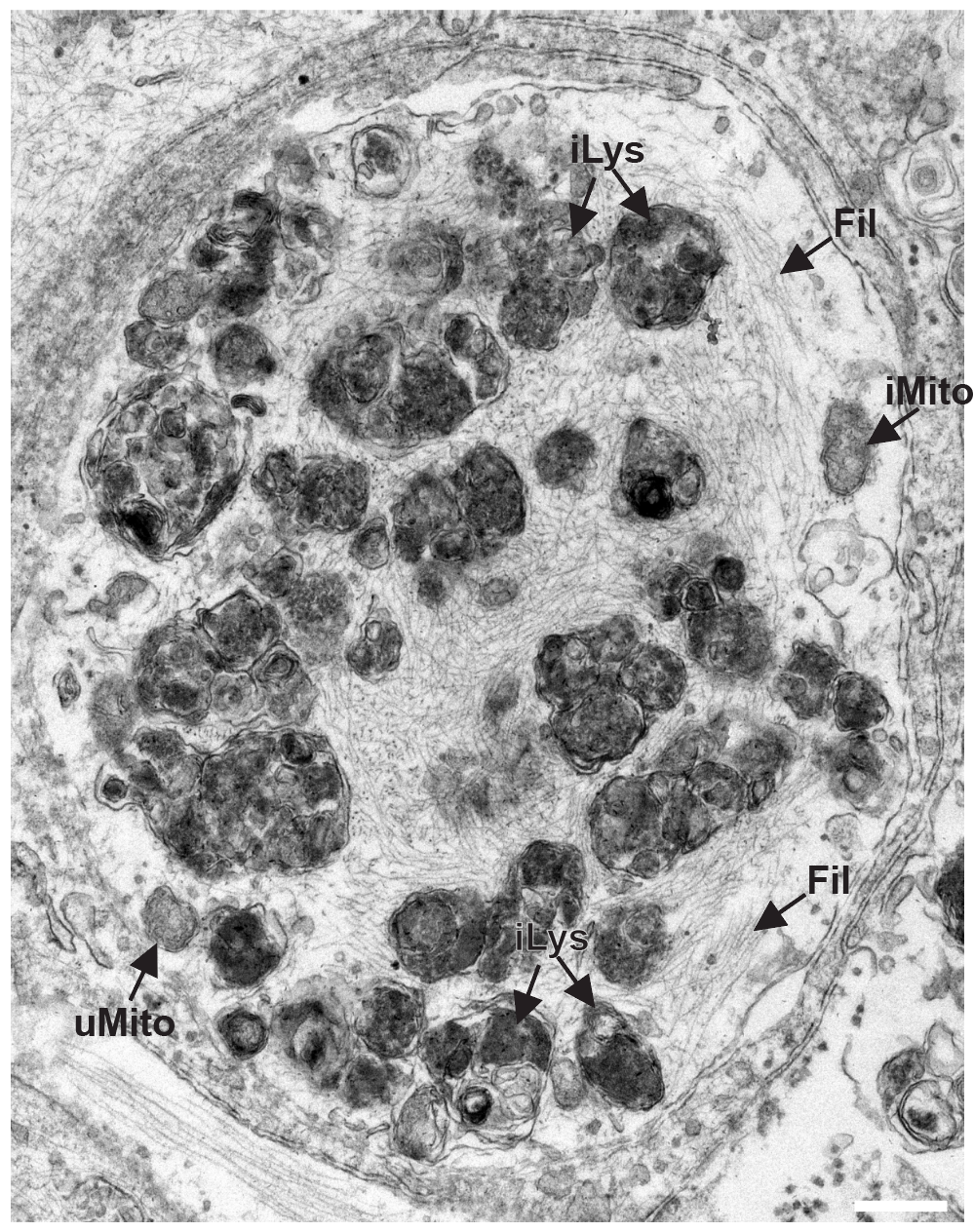
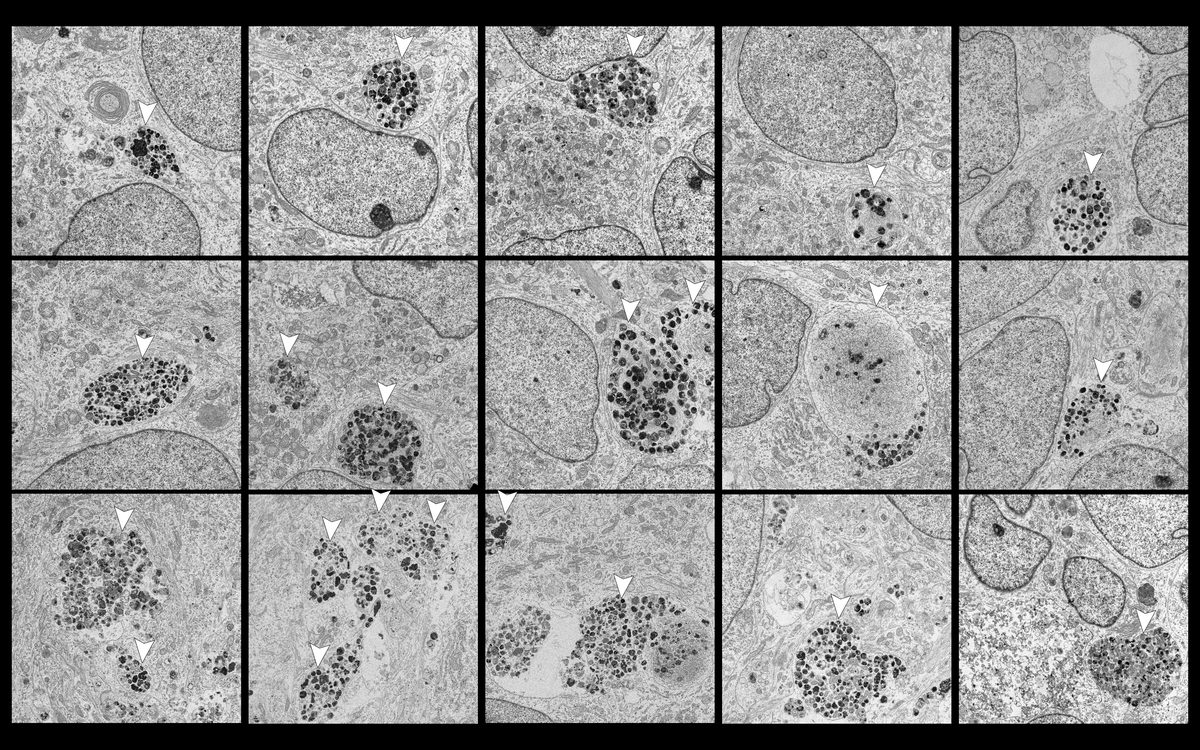
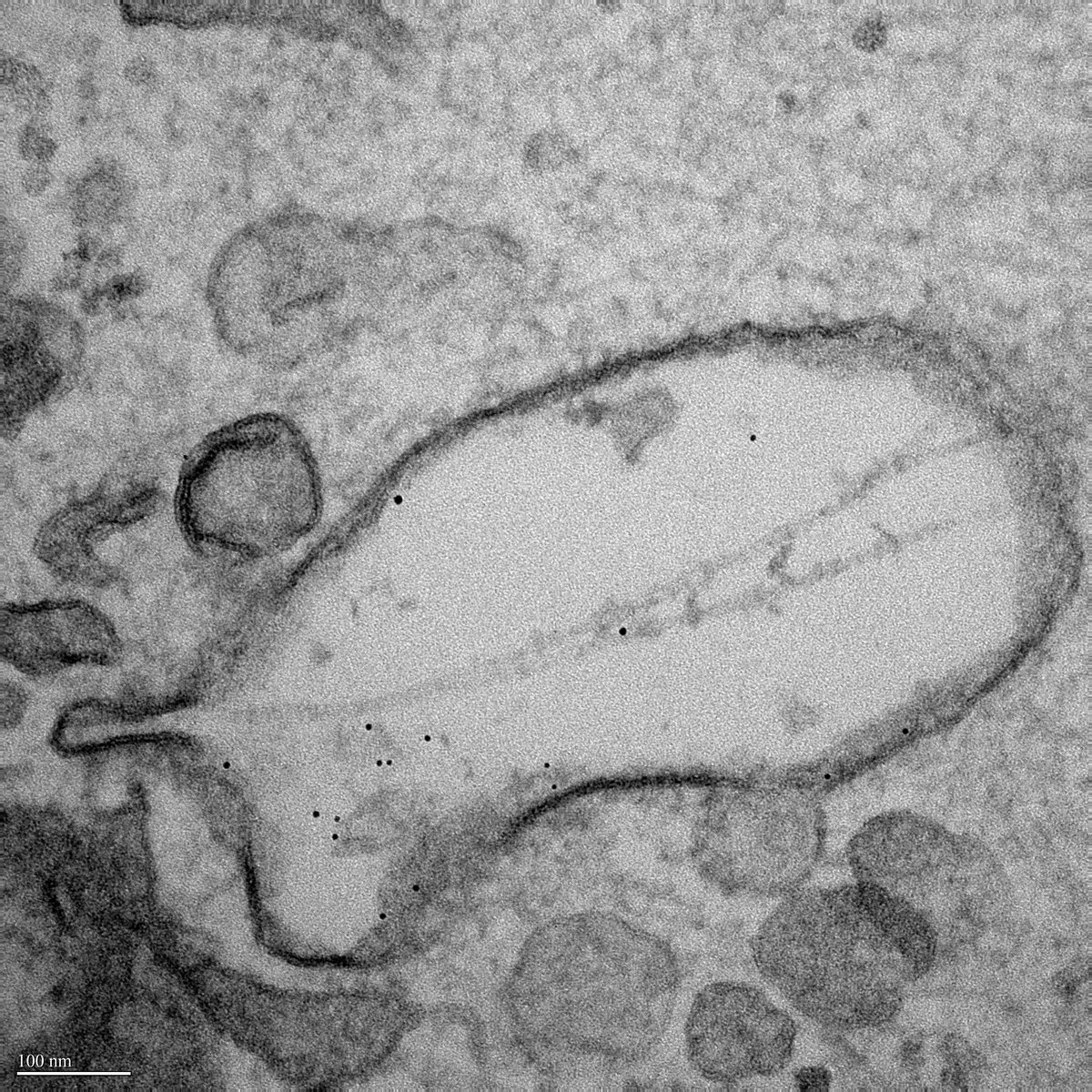
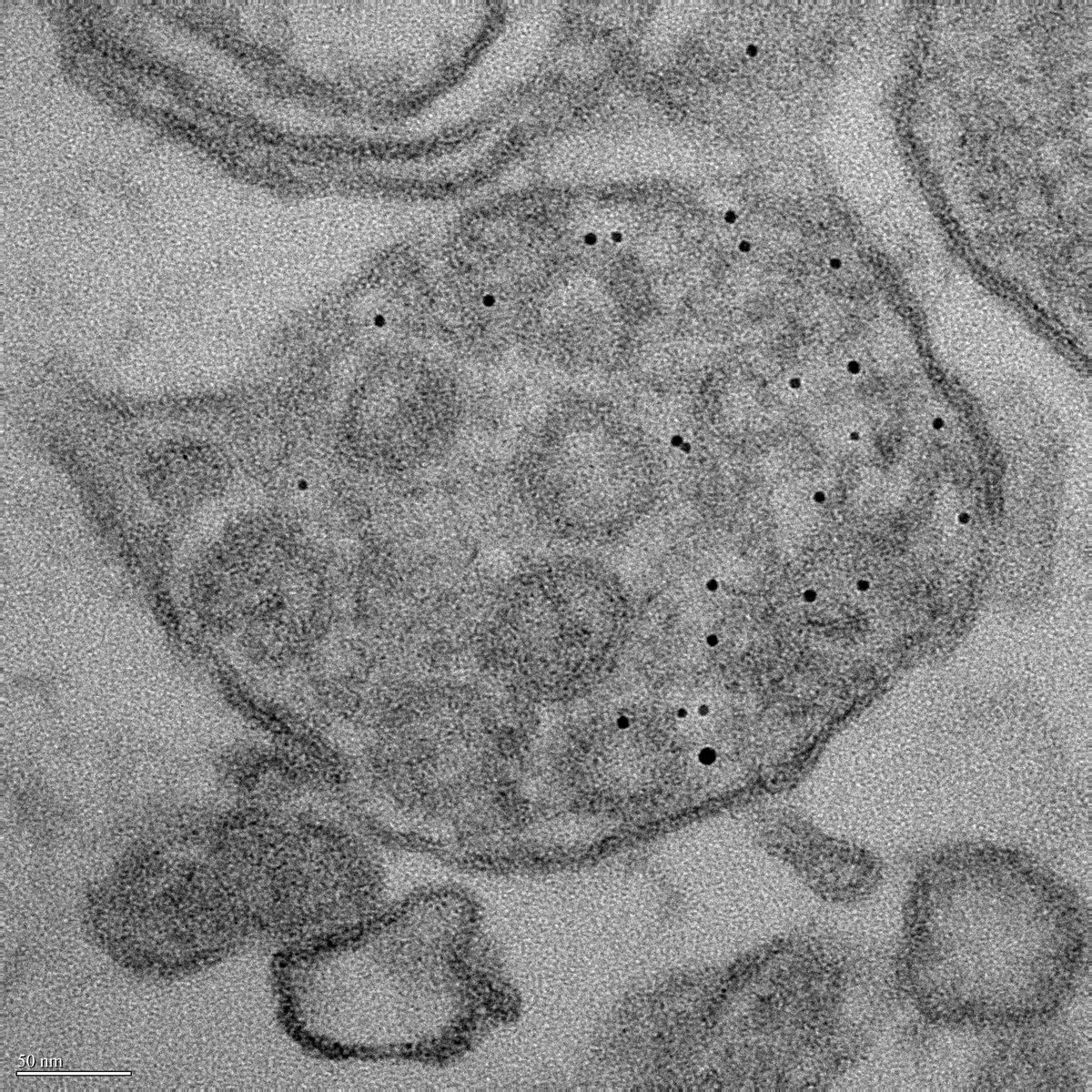
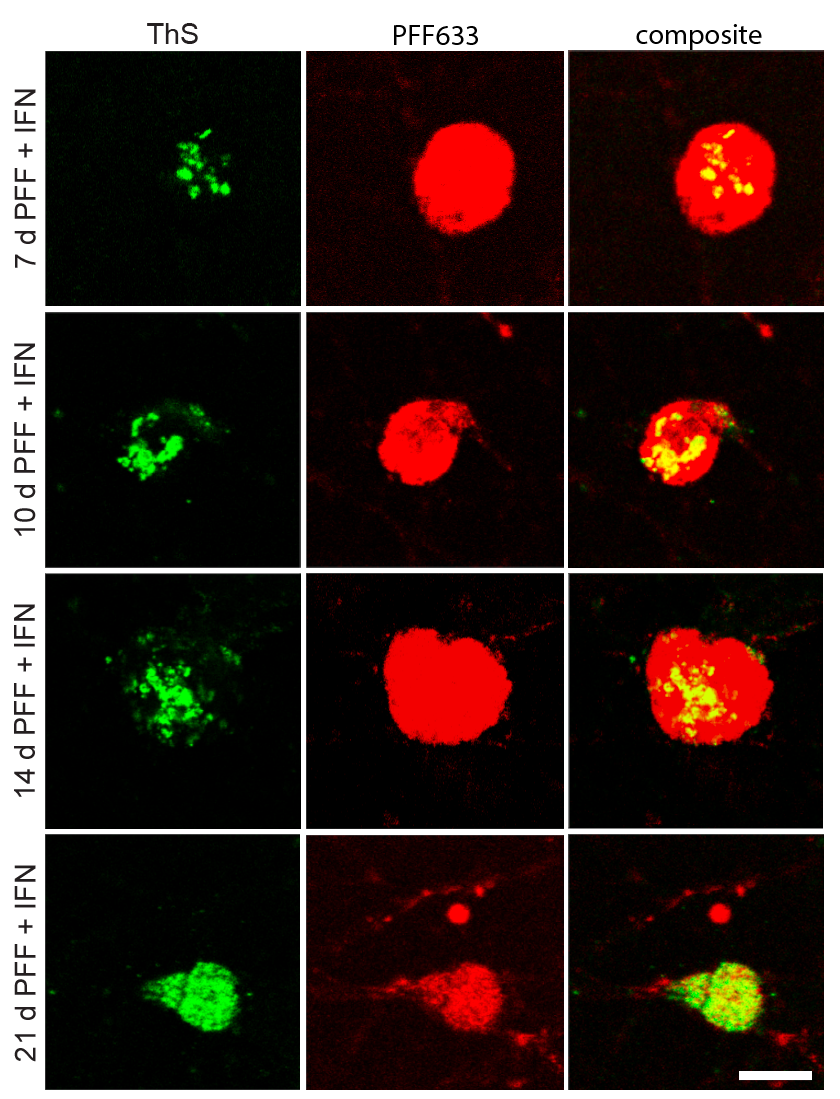
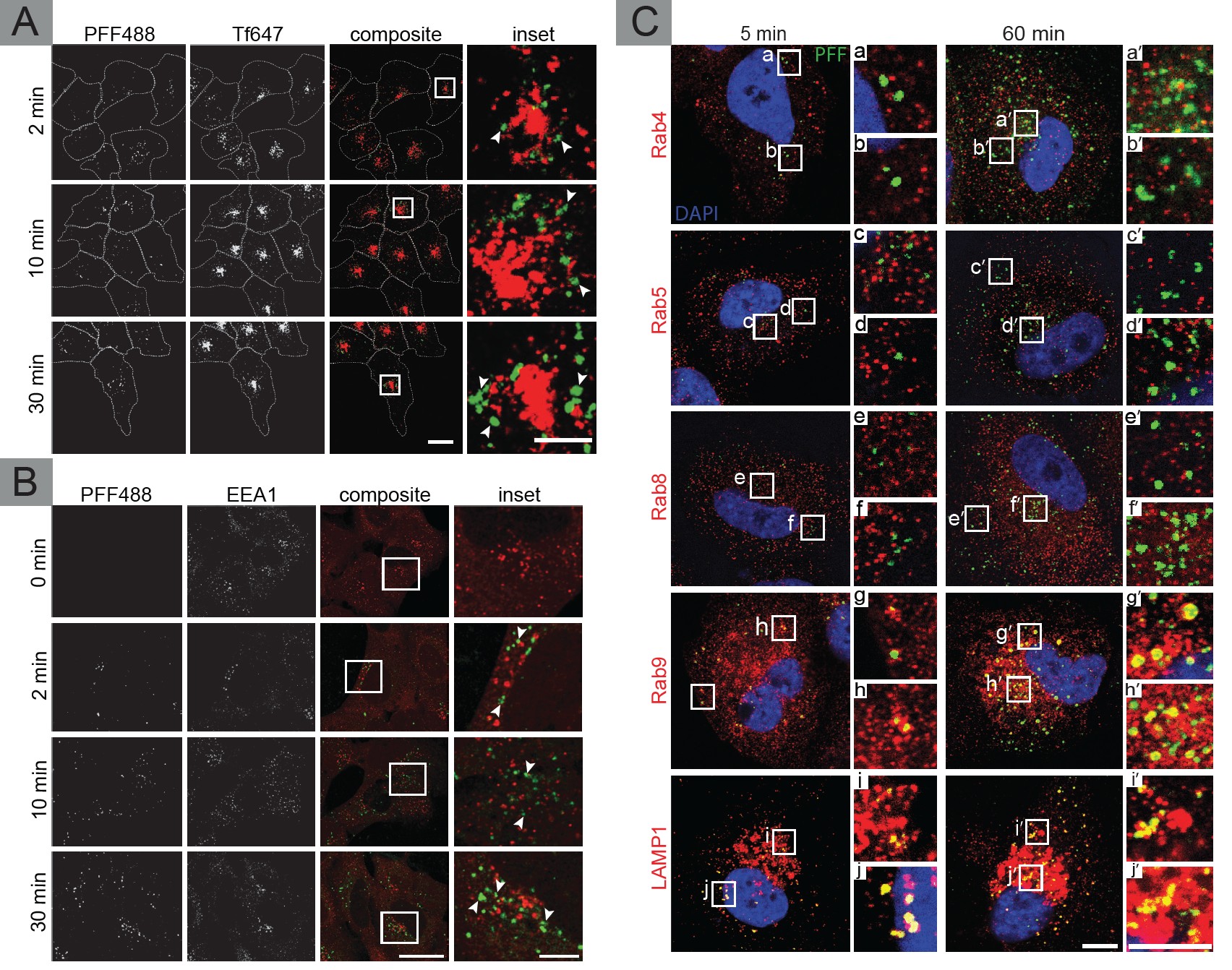
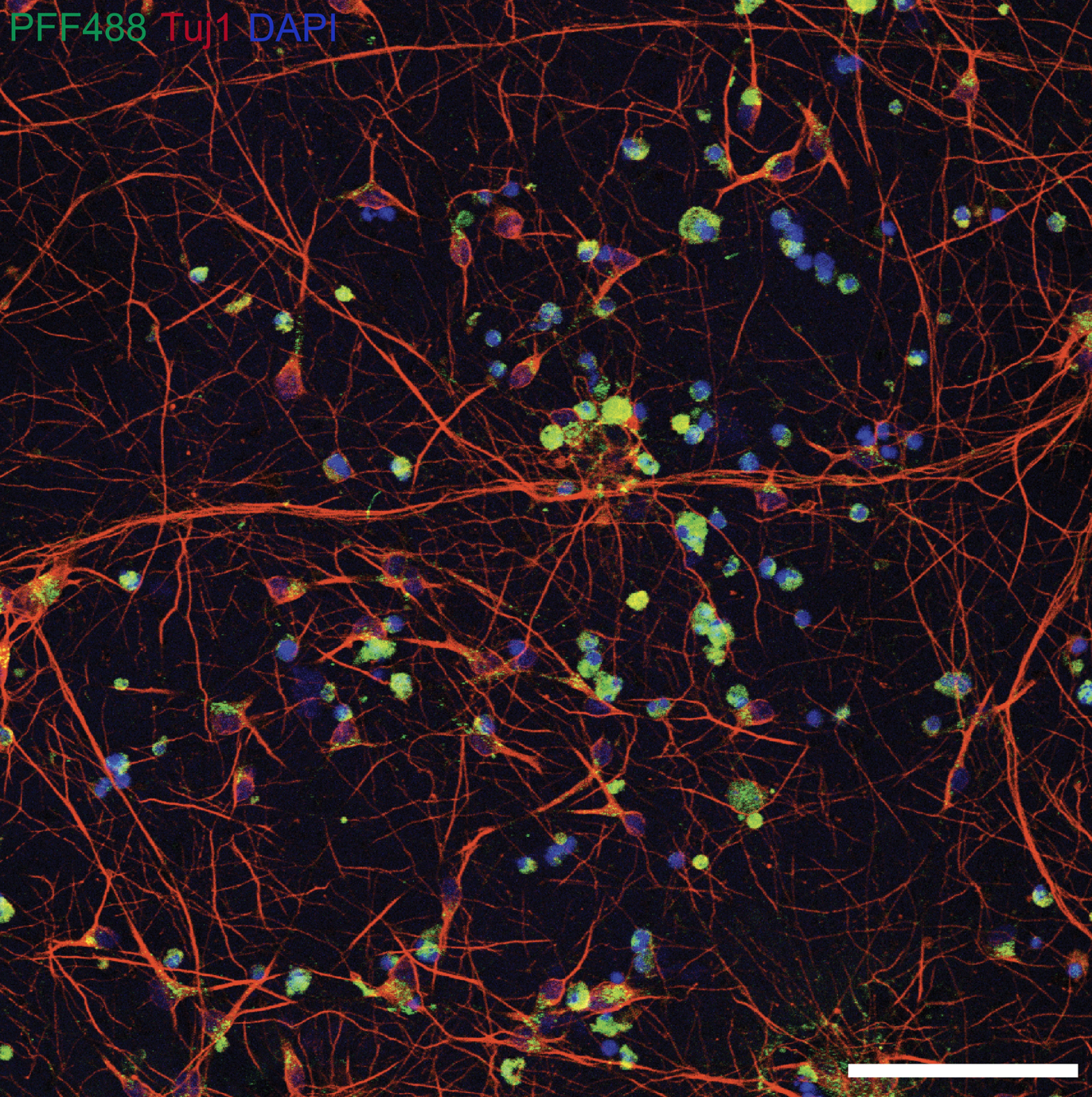
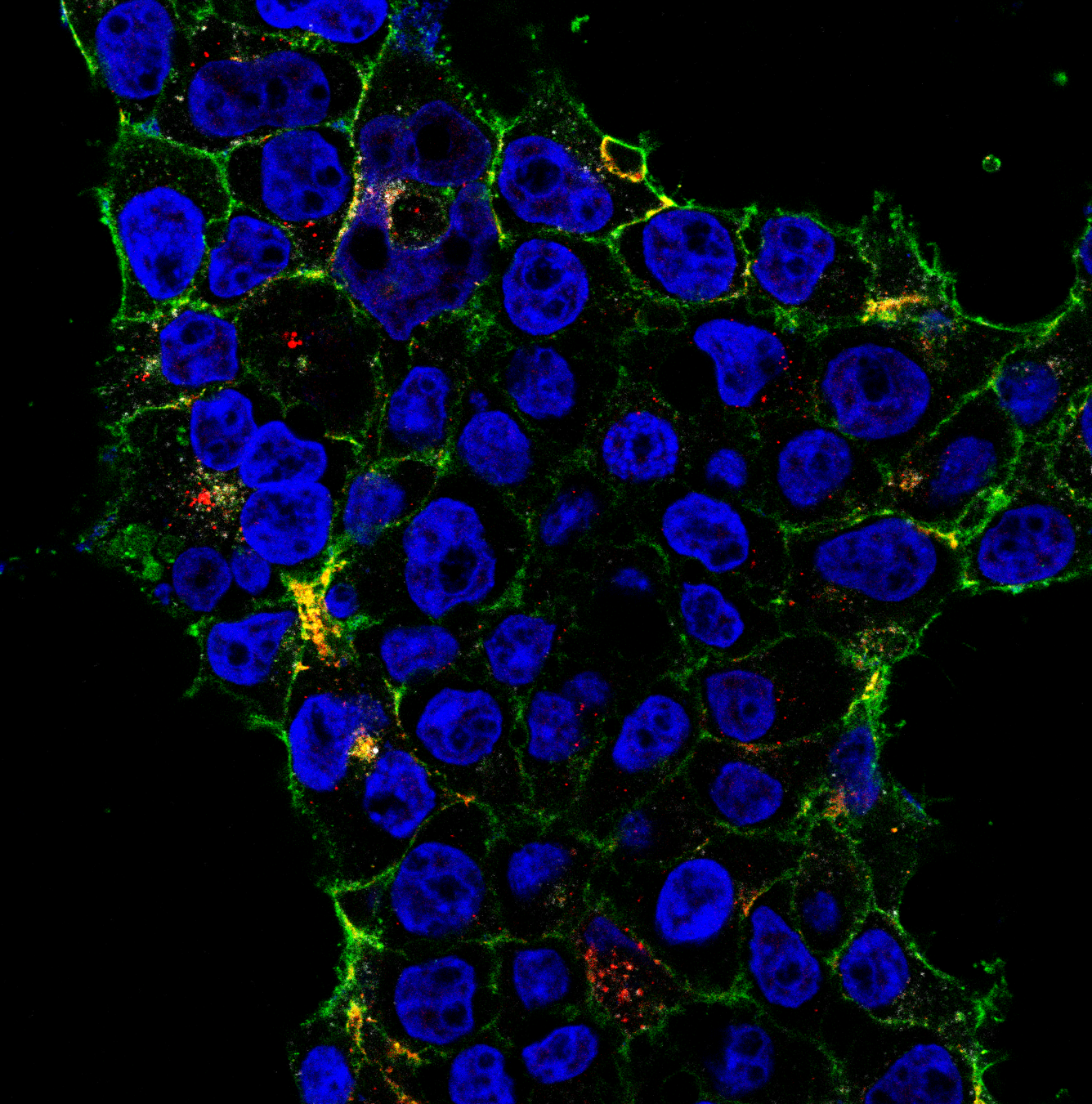
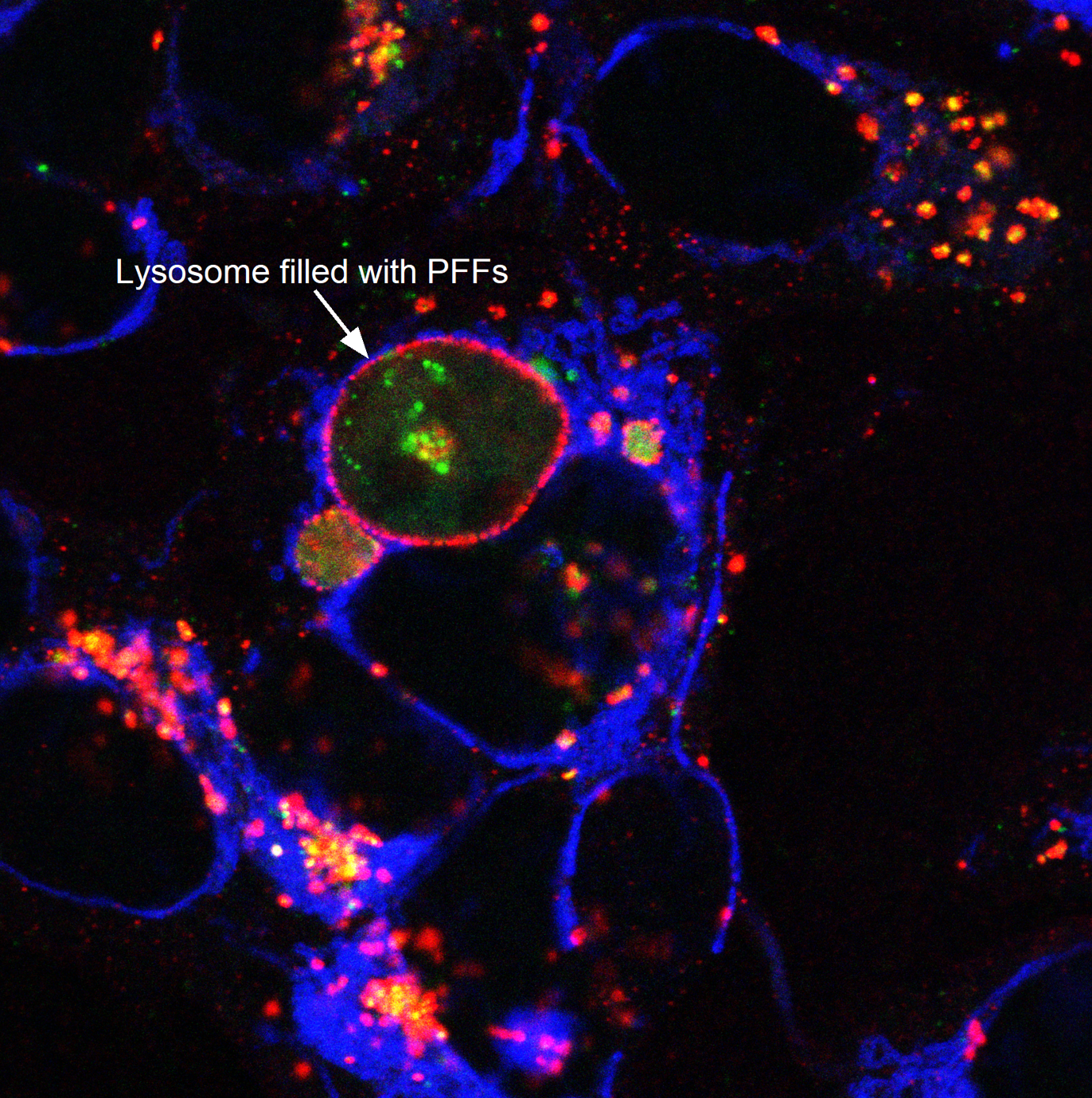
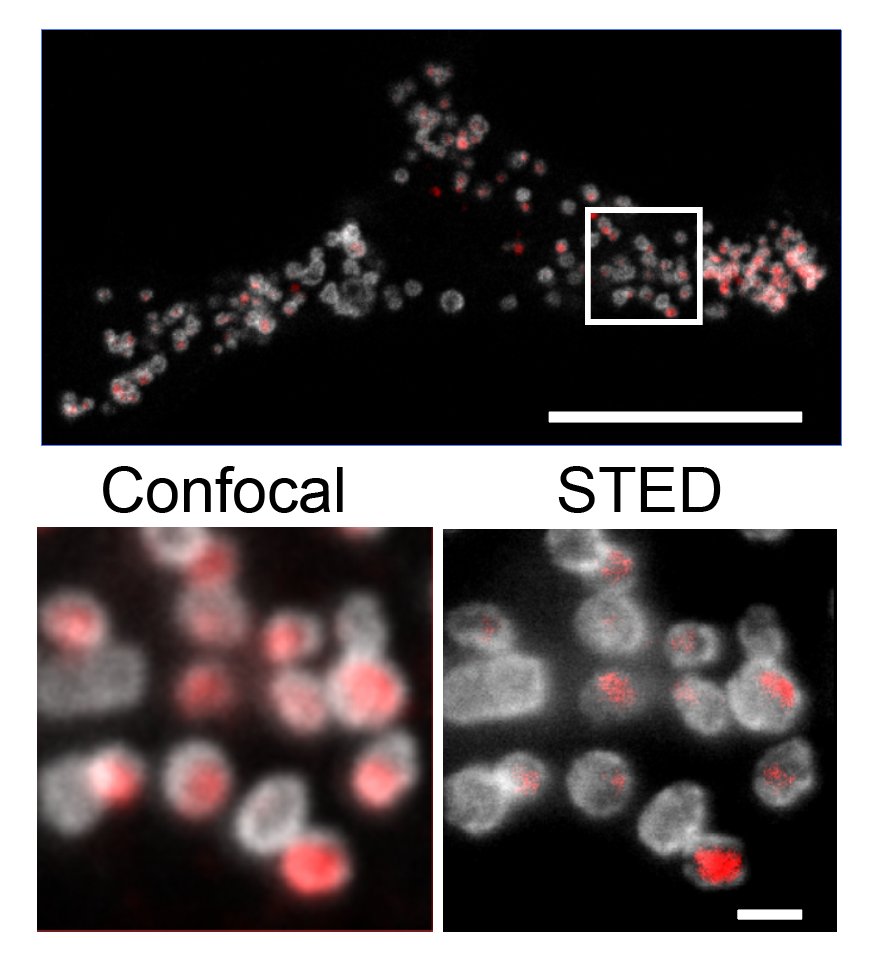
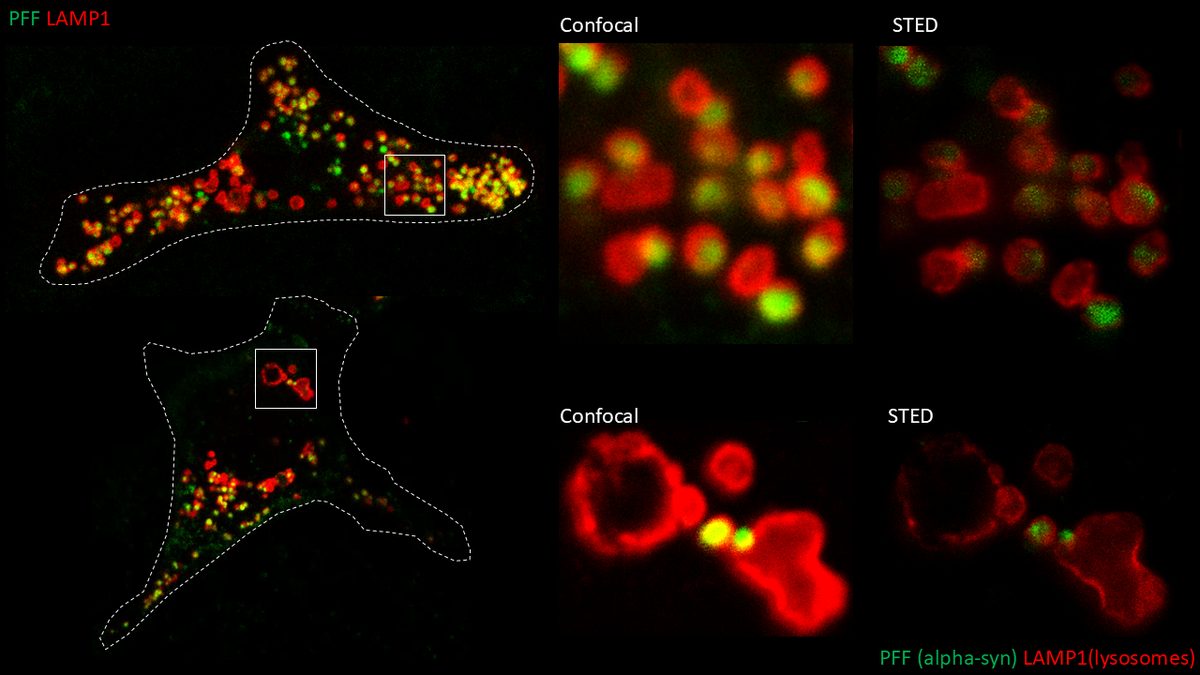
I perform confocal microscopy (Leica) for live and fixed samples, tracking rapid uptake of α-synuclein fibrils and spike protein internalization. STED microscopy (Abberior) resolves sub-lysosomal fibril localization. Zeiss Airyscan provides super-resolution for directional transport and spatial redistribution studies. TIRF (Nikon) for membrane-proximal events. Live-cell imaging captures dynamic processes: fibril internalization, lysosomal swelling, mitochondrial fragmentation, and Parkin translocation.
 1
1
 2
2
 3
3
 4
4
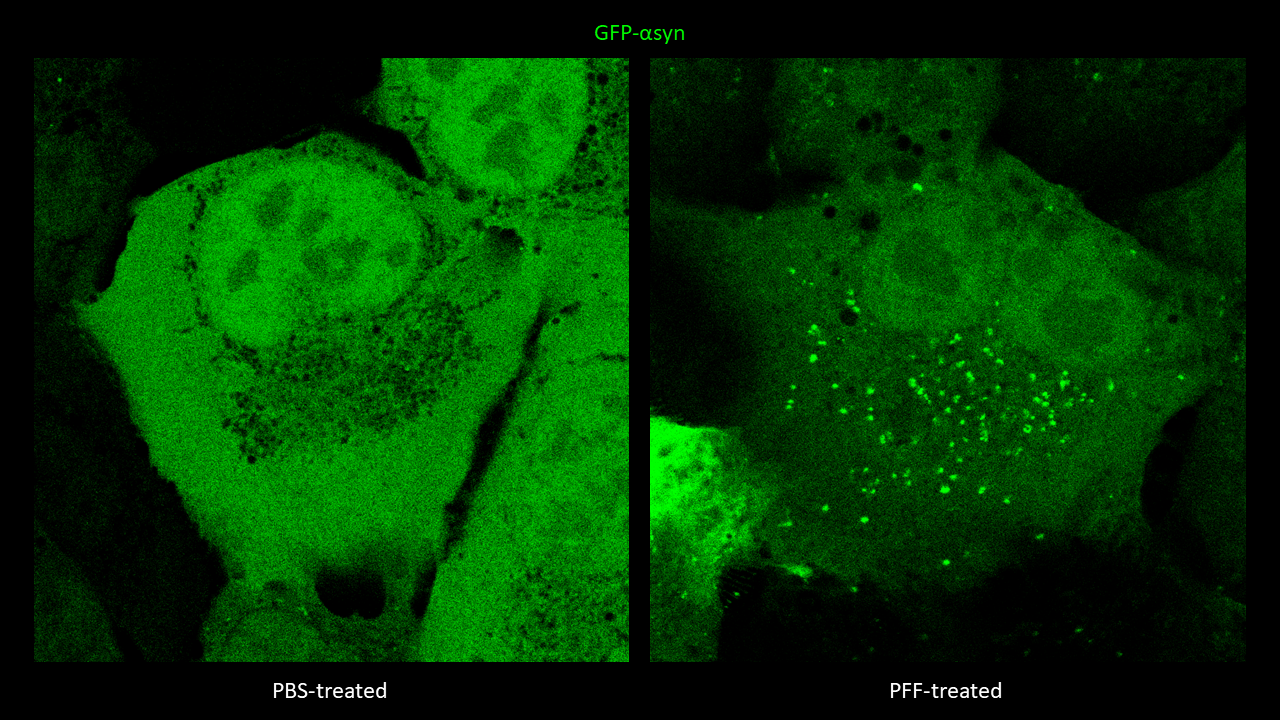 5
5
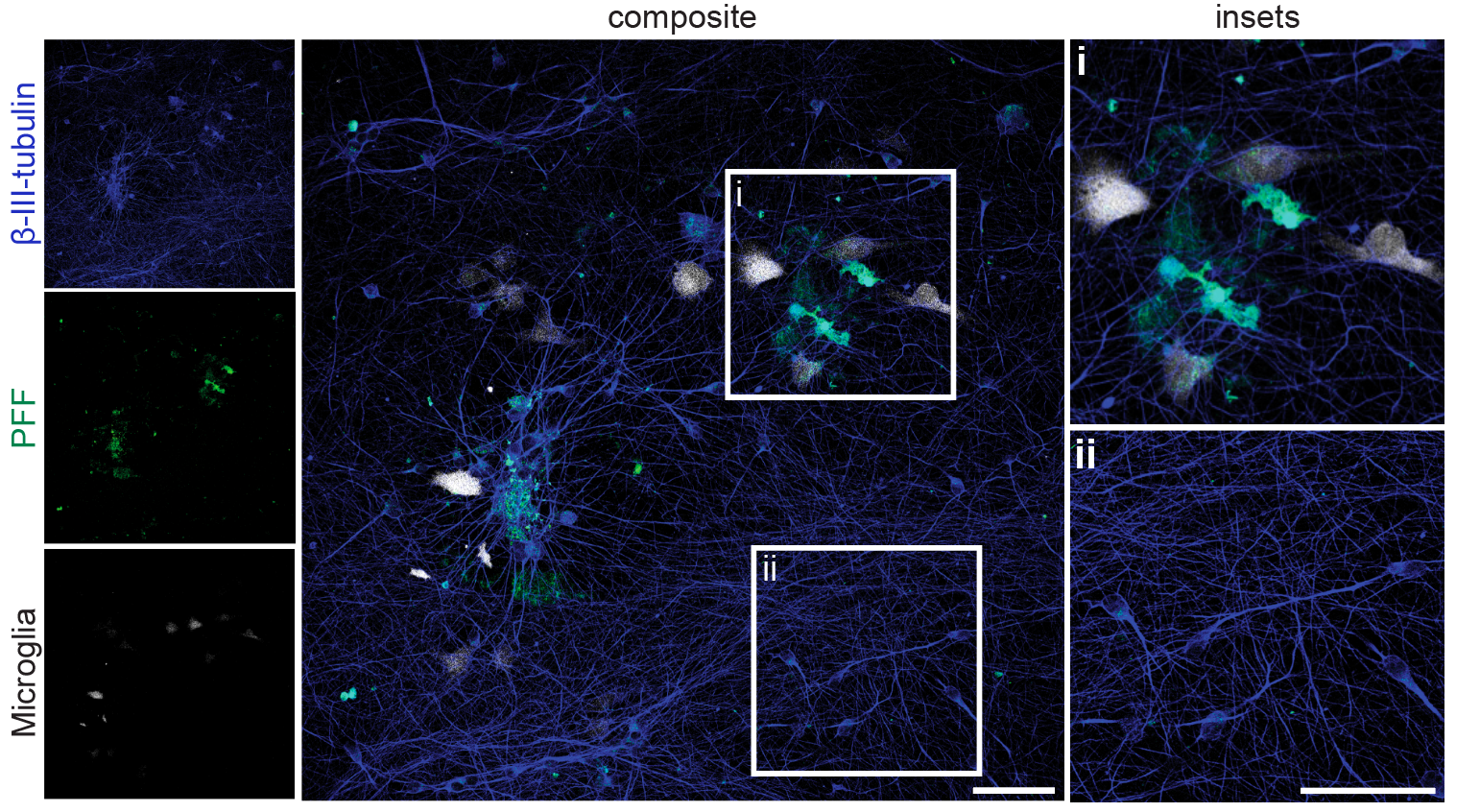 6
6
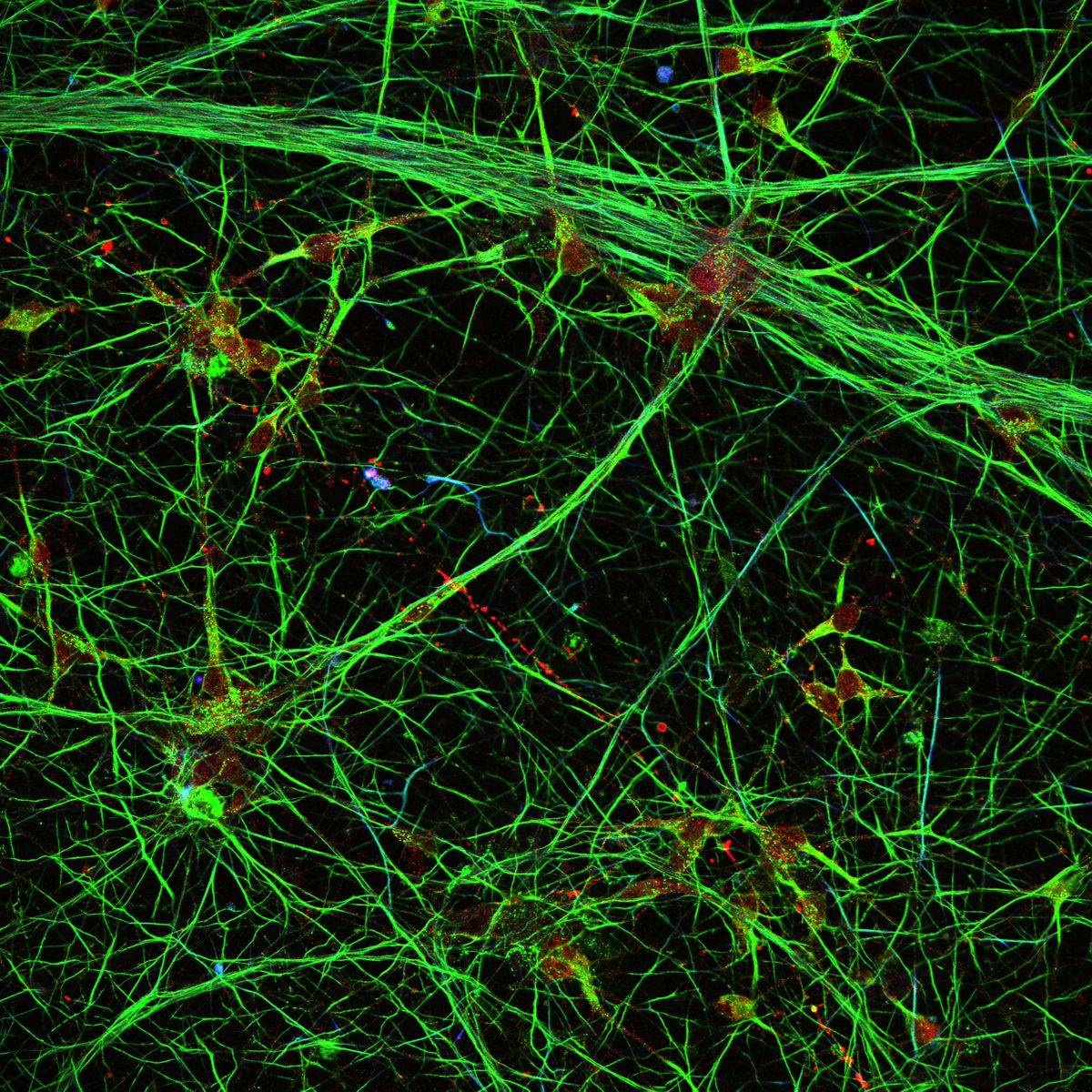 7
7
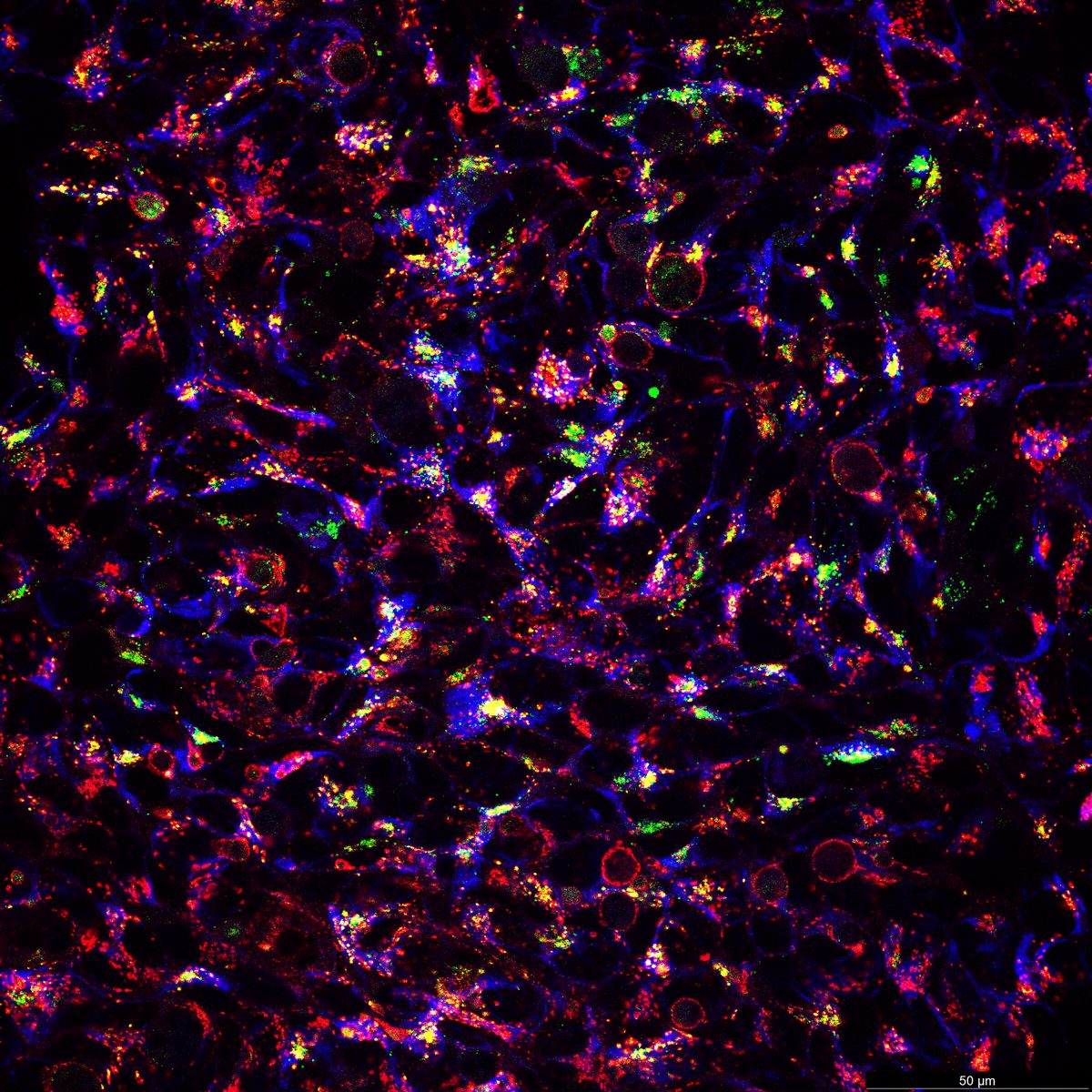 8
8
 9
9