Cytokine-Driven Neuronal Vulnerability
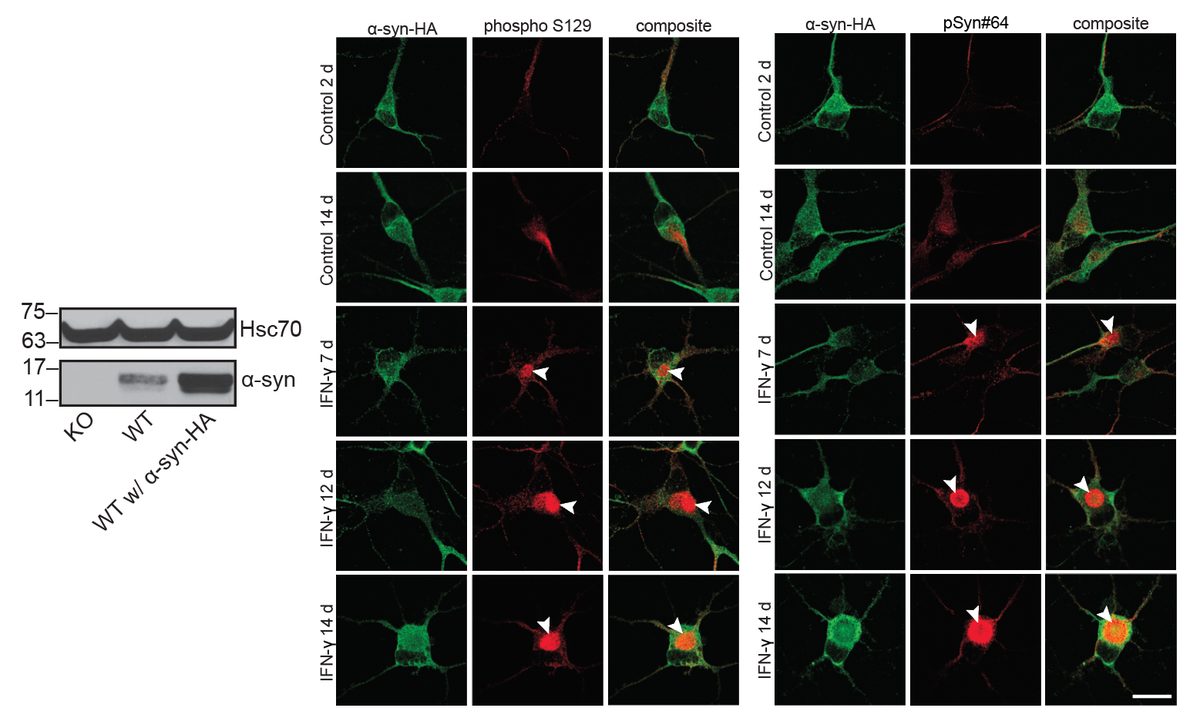
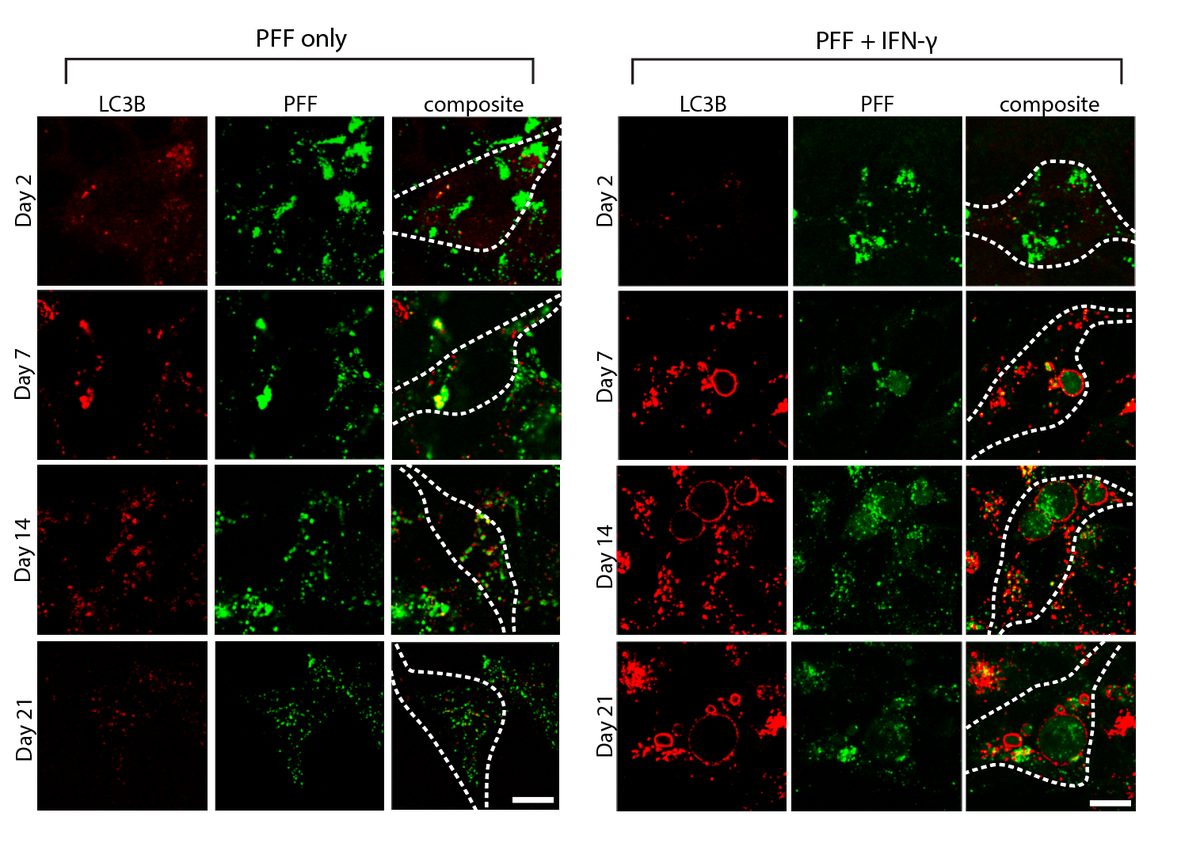
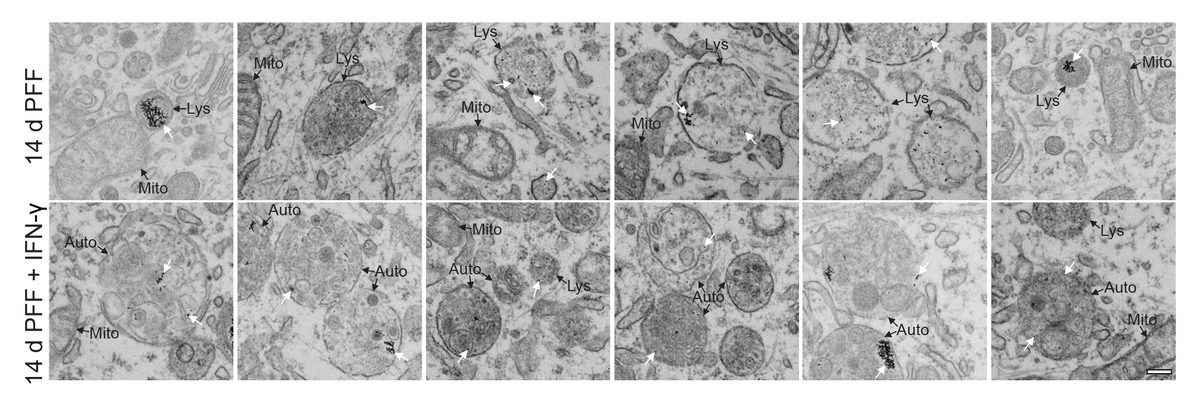
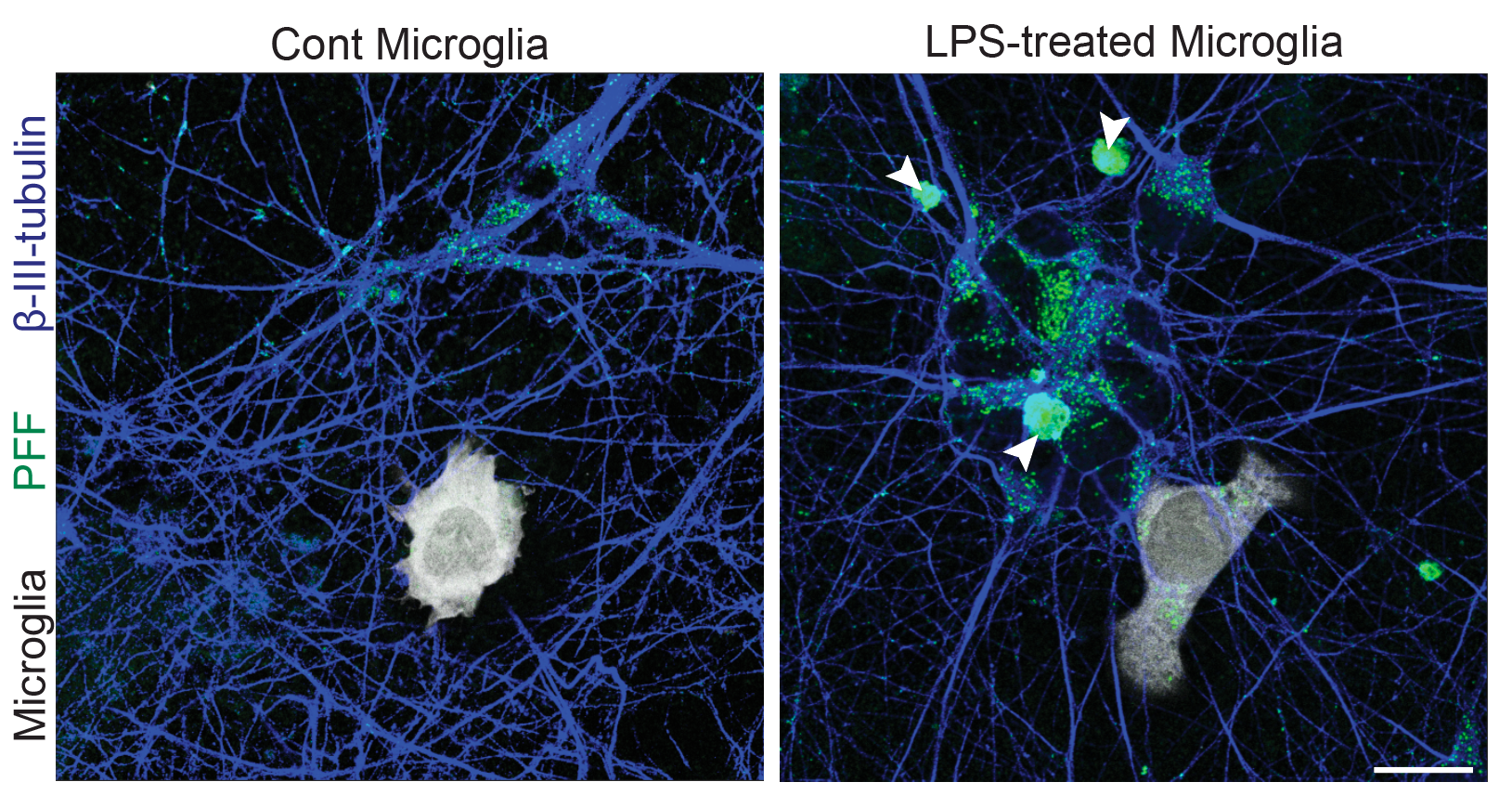
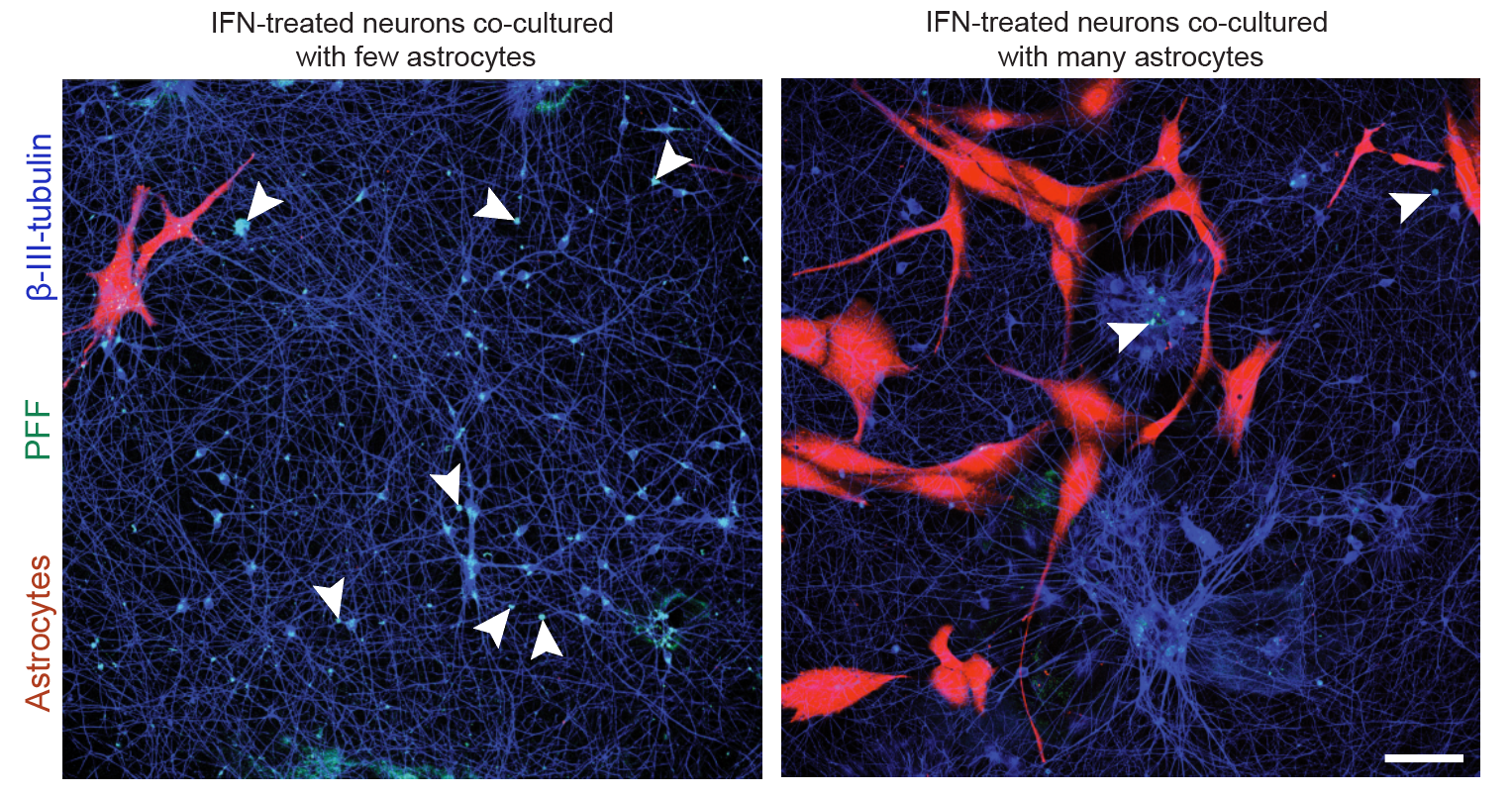
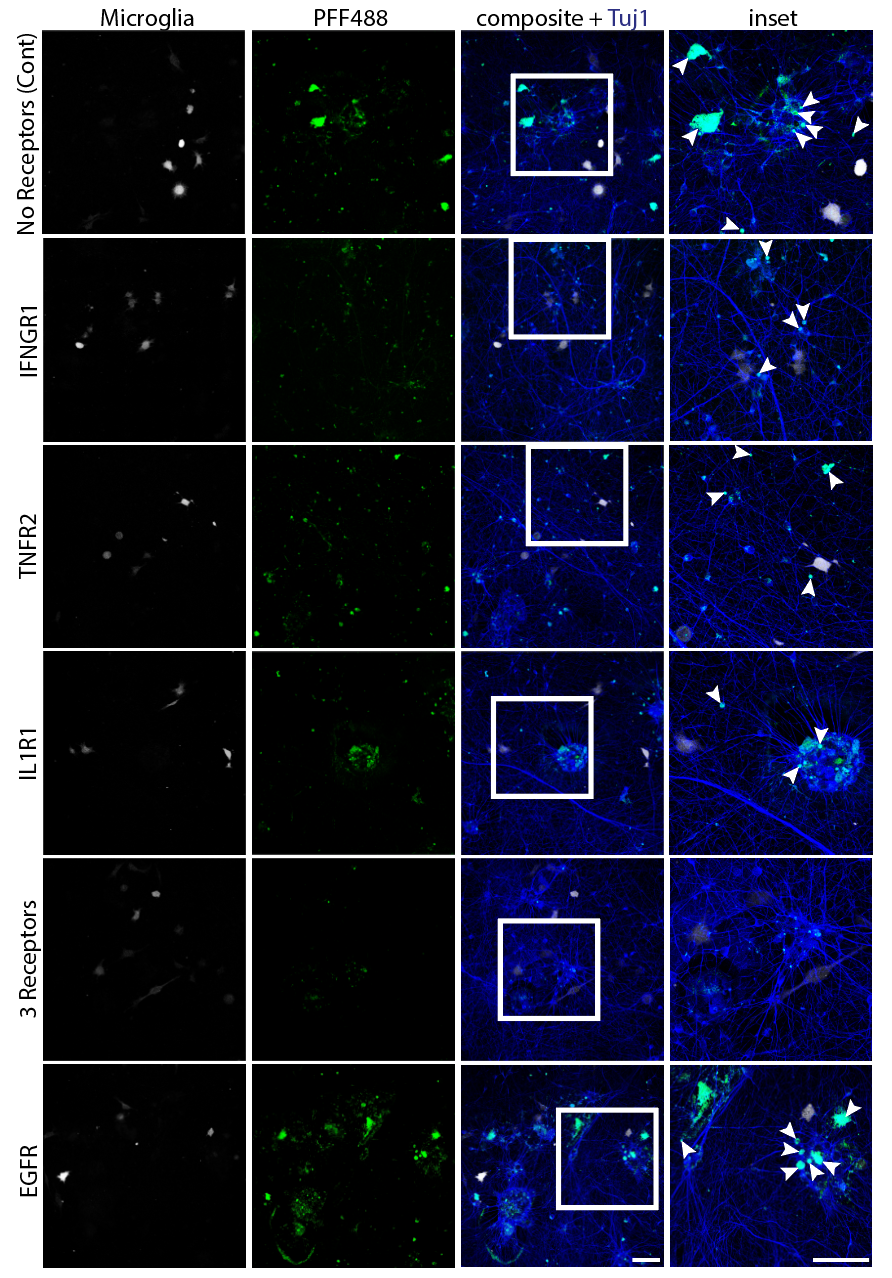
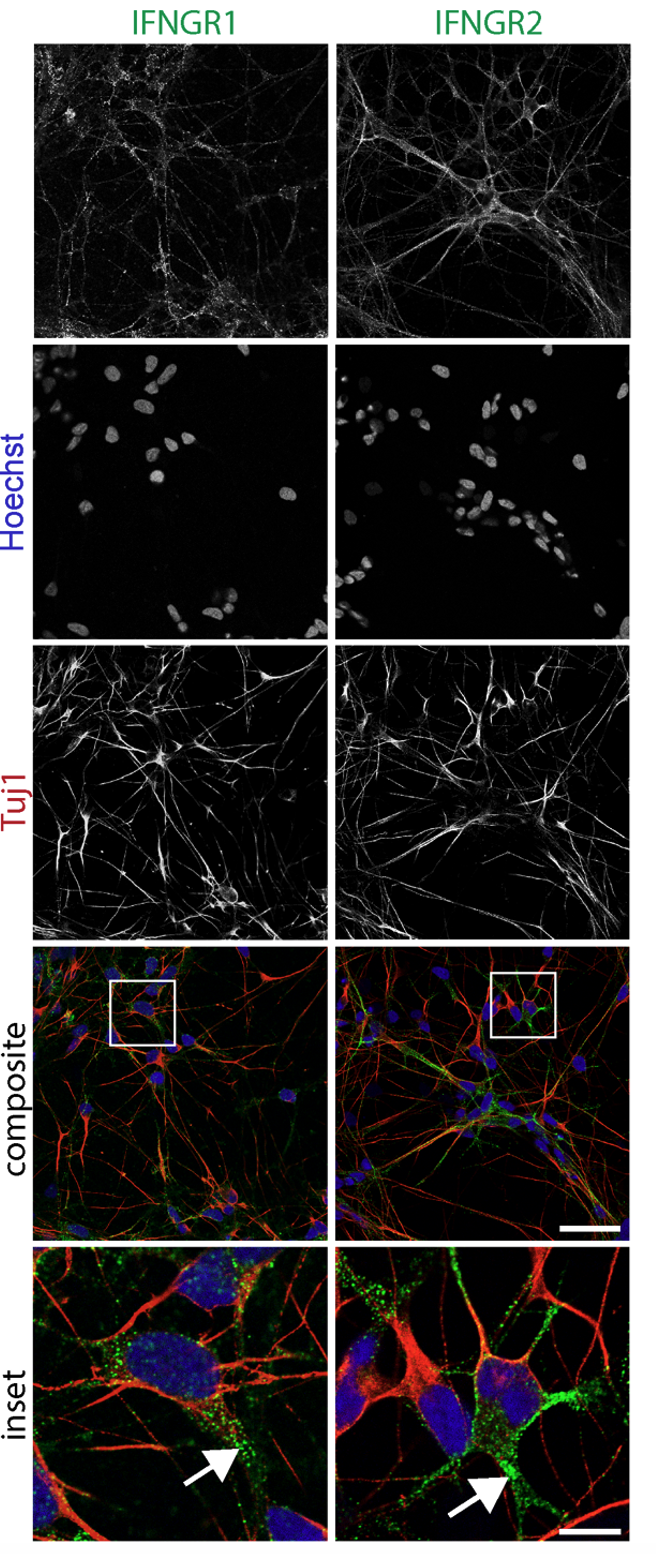
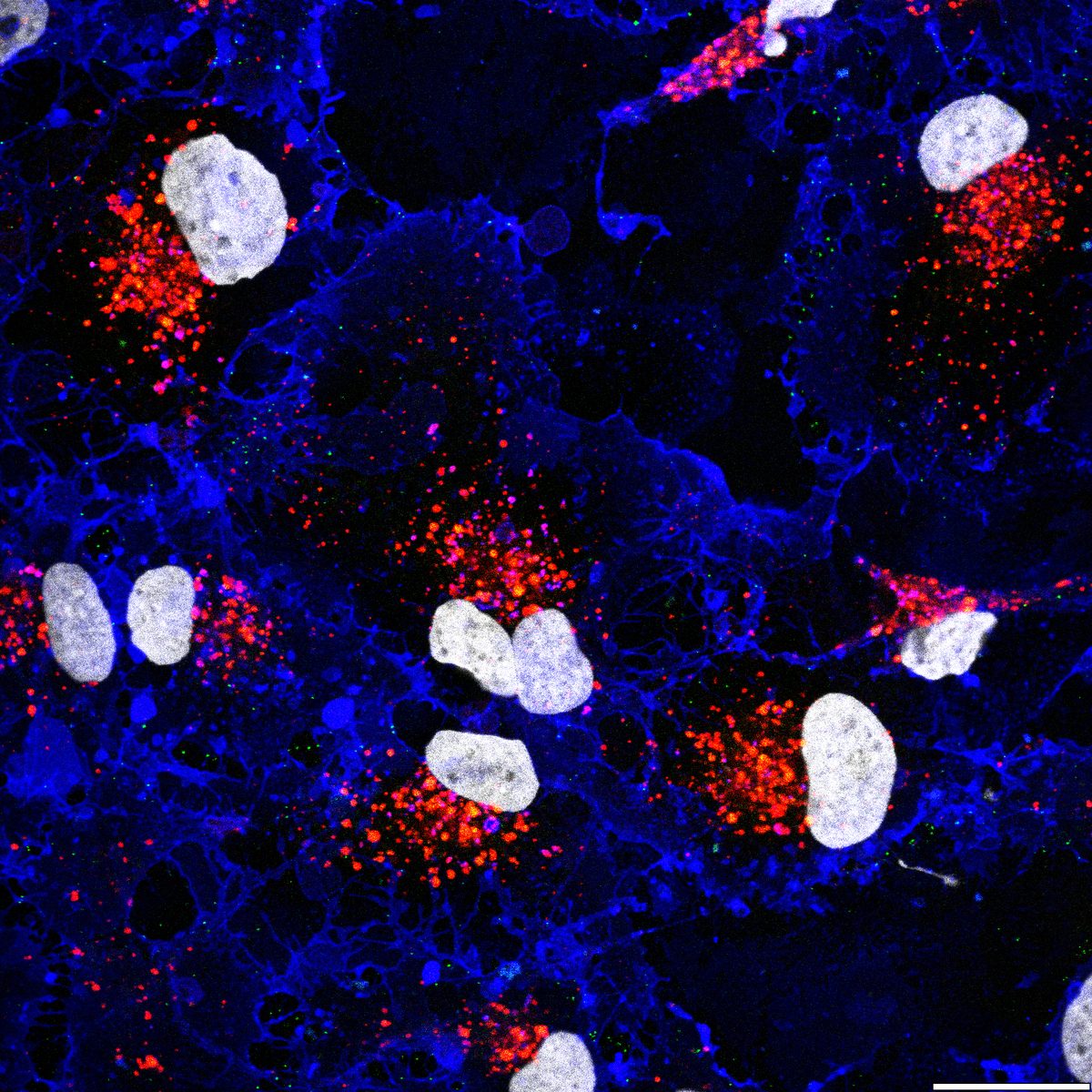
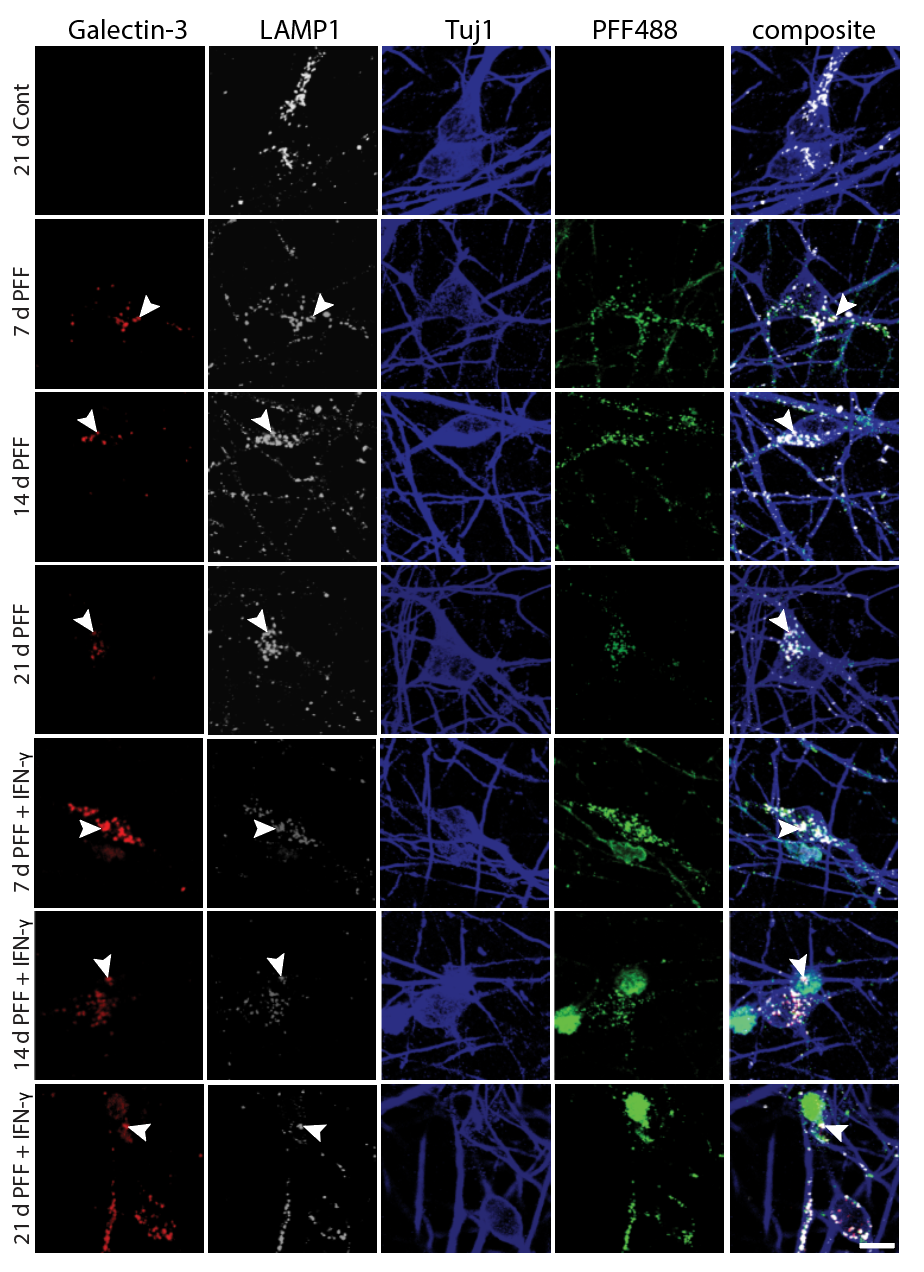
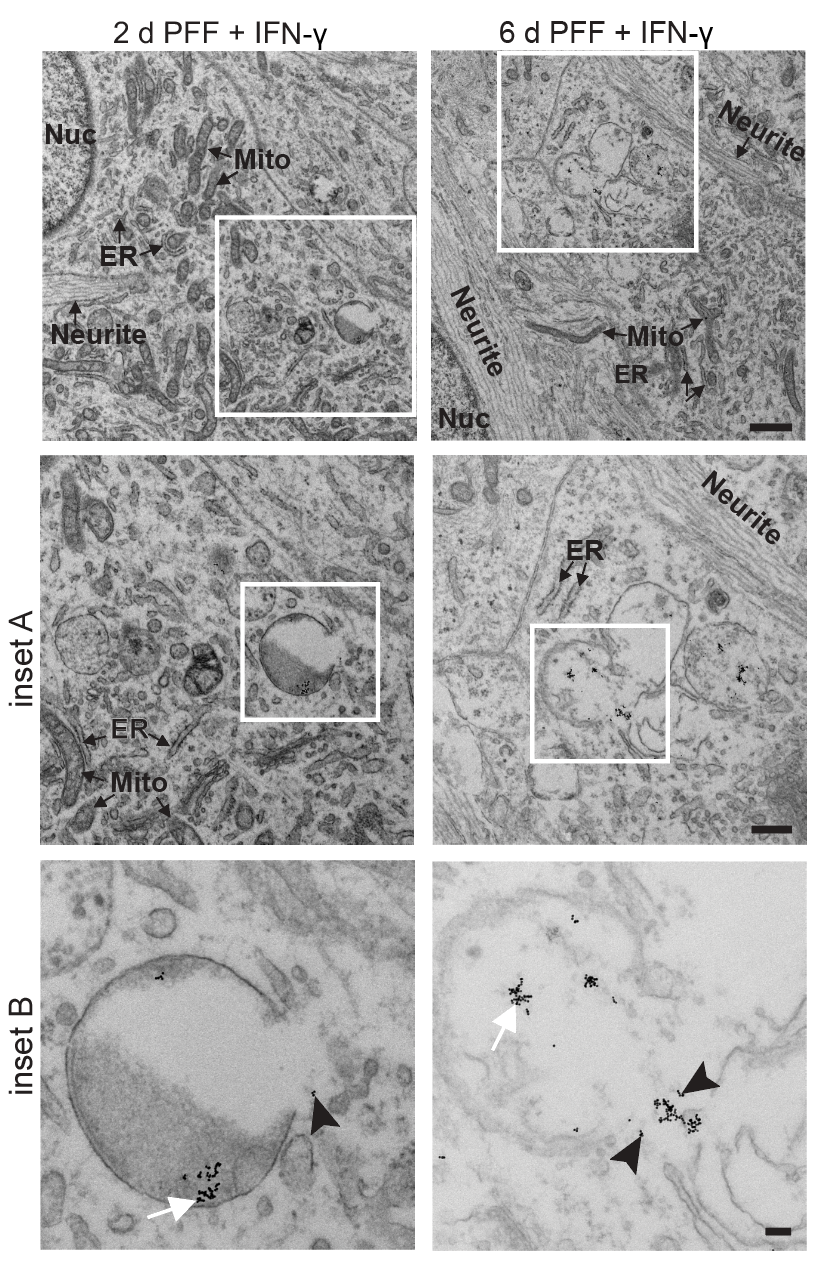
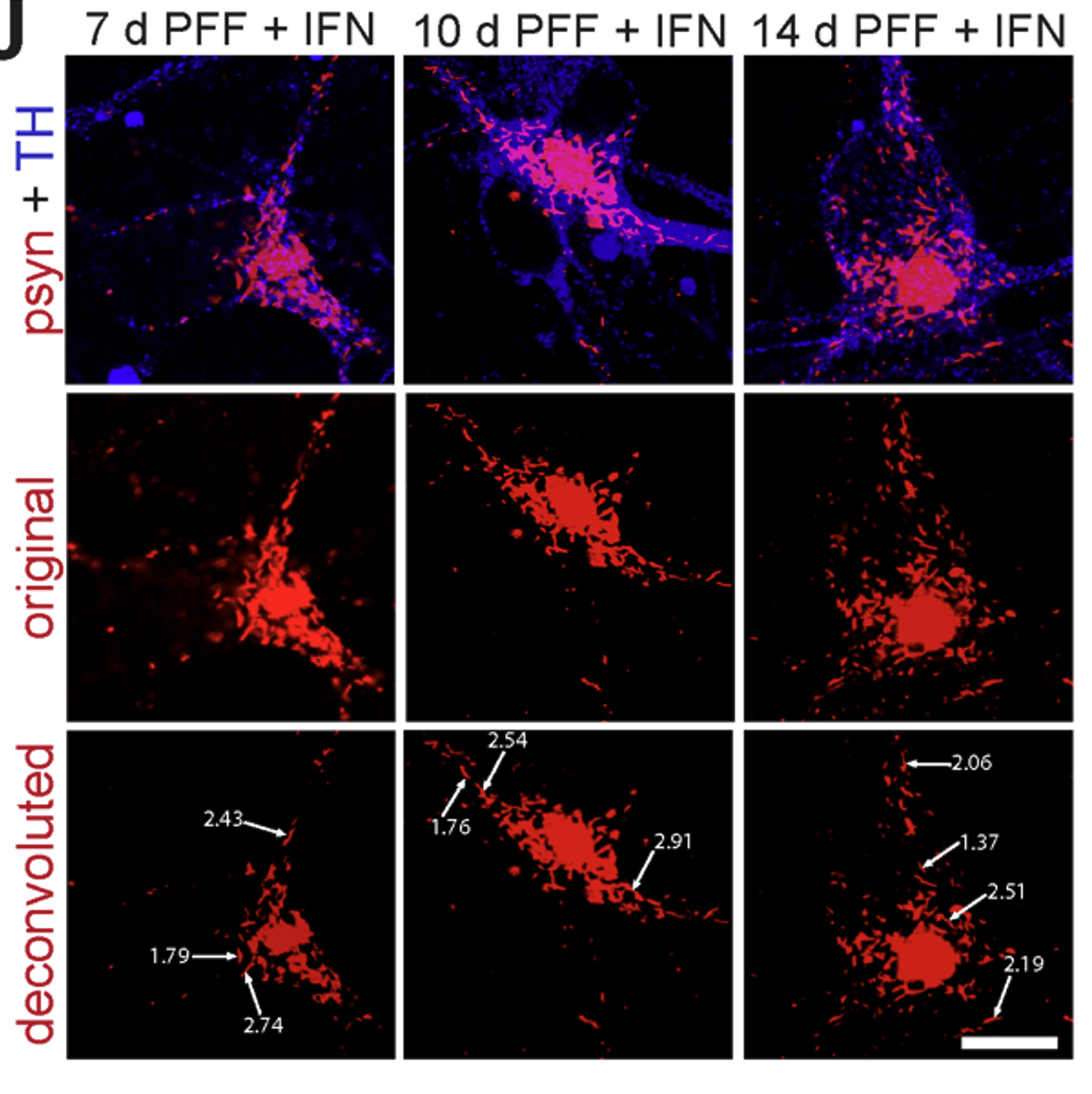
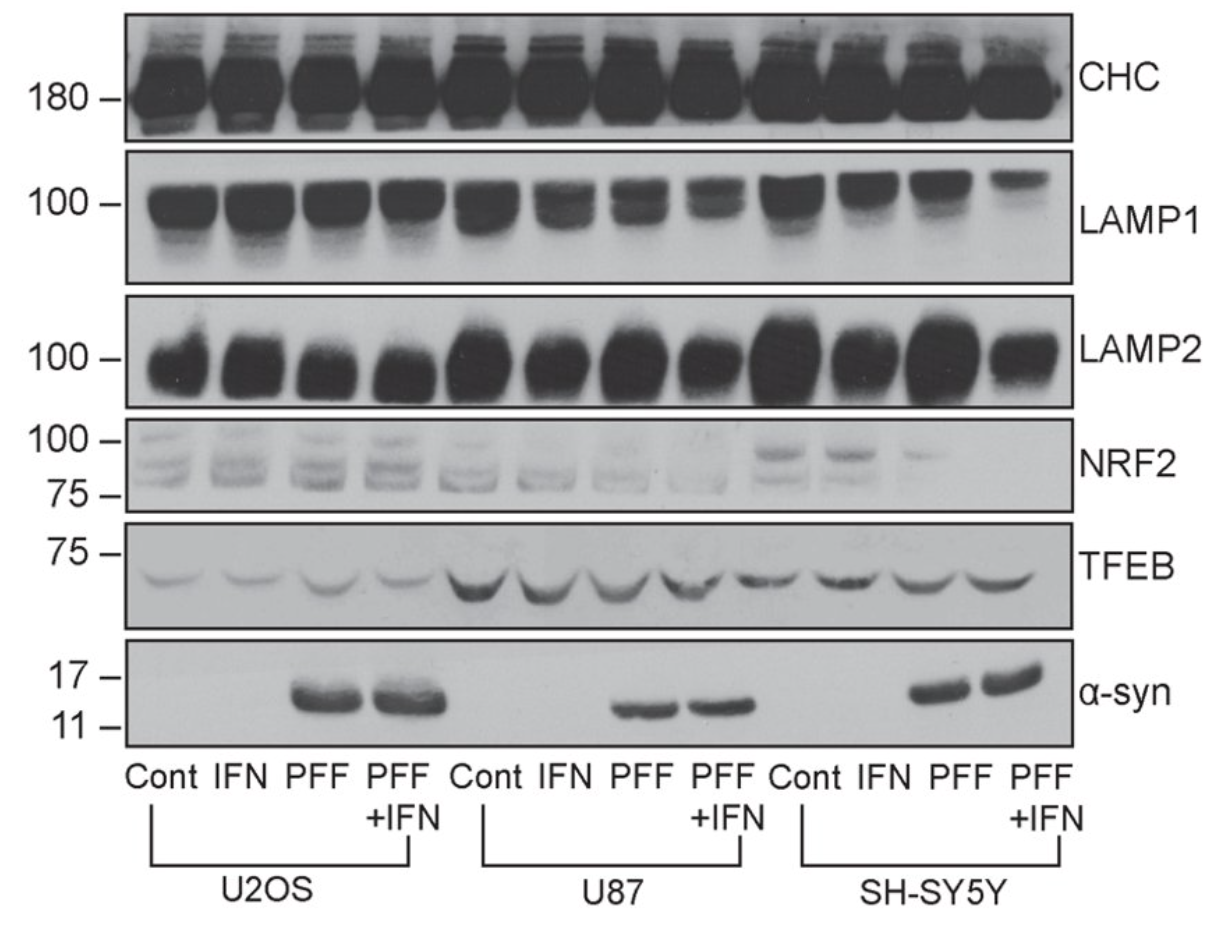
Systematic investigation of how pro-inflammatory cytokines impact iPSC-derived dopaminergic neuron health. IFN-γ exposure induces MHC-I upregulation, antigen presentation machinery activation, and downstream lysosomal dysfunction. TNF-α and IL-1β act synergistically to promote α-synuclein phosphorylation and Lewy body-like inclusion formation. These findings establish a mechanistic framework connecting peripheral inflammation to neuronal pathology in Parkinson's disease.